Understanding air pollution
Understanding the difference between global warming and local air toxicity
1. Introduction
Air emissions are often reduced to a single metric: carbon emissions. While climate change is important, it is only one aspect of air pollution. The atmosphere is a delicate chemical solution, and when businesses introduce foreign agents—whether through combustion, chemical solvents, or logistics—they disrupt a balance that has existed for millions of years.
To manage environmental impact effectively, we must look beyond carbon and understand the broader spectrum of atmospheric degradation.
In this article, you will learn:
✅ Why the air is important and how it impacts us
✅ The difference between the greenhouse gas emissions and air pollution
✅ Pollution traveling thousands of kilometers
✅ How specific industrial activities alter the air
By the end of this article, you will have a better understanding of air pollution.
1. Air, a dynamic mixture
Before understanding pollution, we must first appreciate the delicate baseline from which we are deviating. The Earth’s atmosphere is not merely the space between us and the stars; it is a complex, dynamic, and thin protective envelope that makes life possible. Think of the atmosphere as Earth’s life-support system. It acts like a shield (blocking harmful solar rays), a blanket (keeping us warm), and a water delivery system (moving rain around the world).
The perfect mixture
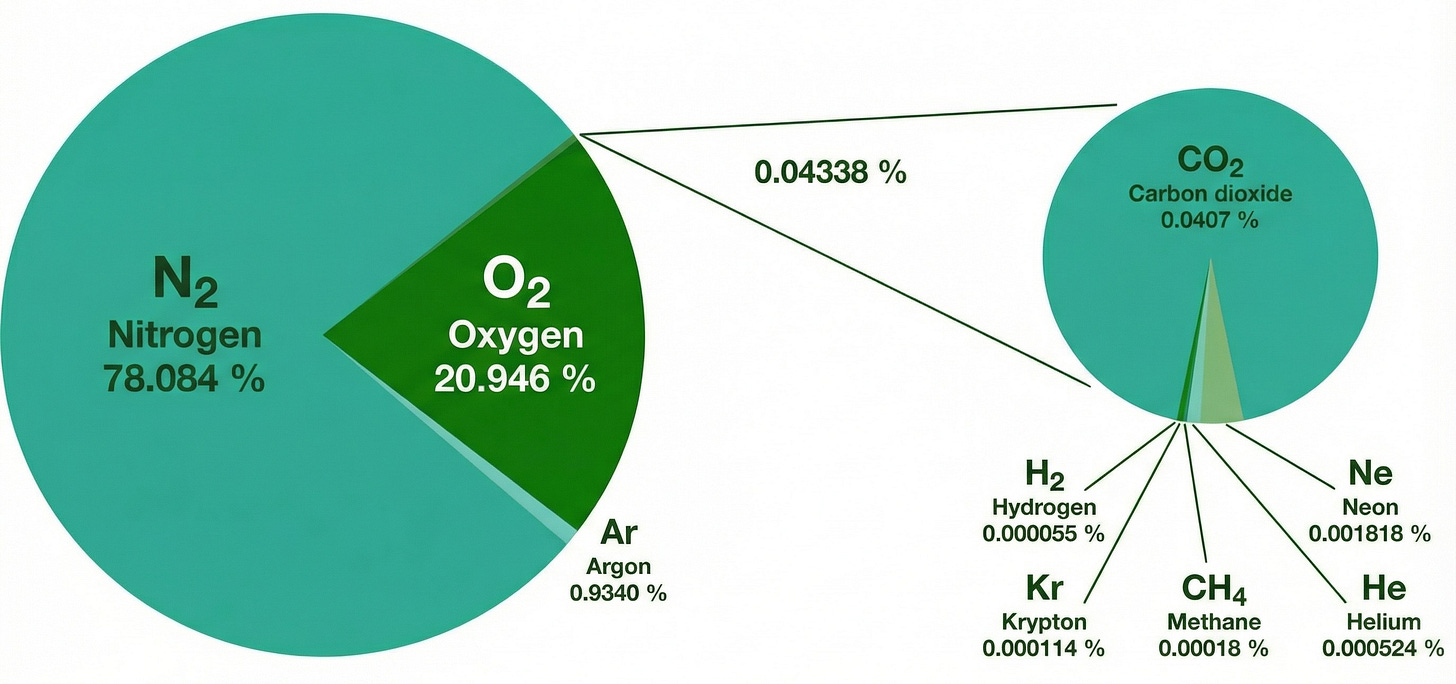
For millions of years, the atmosphere has maintained a relatively stable chemical composition. This specific mix of gases is what biological life has evolved to breathe and thrive in. If you were to take a dry sample of the air we breathe, it would consist of a distinct mixture:
Nitrogen (~78%): The most abundant gas and essential for building proteins in plants and animals via nitrogen fixation in the soil.
Oxygen (~21%): The fuel for respiration in most living organisms and a key component in the chemical reactions that drive our ecosystems.
Argon (~0.9%): A noble gas that is chemically inert.
Trace gases (~0.1%): This tiny fraction includes naturally occurring Carbon Dioxide, Neon, Helium, Methane, and water vapor.
The power of the 0.1%
It is important to recognize that although trace gases make up less than 0.1% of the atmosphere, they exert a massive influence on the planet’s climate and weather patterns. A slight alteration in this fractional percentage can trigger global shifts in temperature.
Ecosystem services of the atmosphere
Beyond simply providing air to breathe, the atmosphere performs critical services that maintain the planet’s habitability:
Thermal regulation: through the natural greenhouse effect, trace gases trap just enough solar heat to keep Earth’s average temperature at a habitable level (roughly 15°C/59°F). Without this, Earth would be a frozen rock.
Radiation shielding: High in the stratosphere, the Ozone layer acts as natural sunscreen, absorbing the majority of the sun’s harmful ultraviolet (UV) radiation before it reaches the surface.
Meteorological distribution: The movement of air masses redistributes heat and moisture from the equator toward the poles, creating the weather cycles and precipitation patterns required for agriculture and fresh water.
A delicate balance
In its pristine state, the atmosphere exists in dynamic equilibrium. This means that while gases are constantly being exchanged—oxygen produced by photosynthesis, carbon dioxide released by respiration—the overall ratios remain steady. The system is resilient, but it has limits.
When we view the atmosphere not as an infinite dump, but as a finite, delicately balanced chemical solution, the severity of introducing foreign agents becomes clear.
2. Understanding air pollution
Air pollution occurs when the atmosphere’s delicate chemical balance is disrupted by the introduction of harmful solids, liquids, or gases in concentrations that exceed natural capacities.
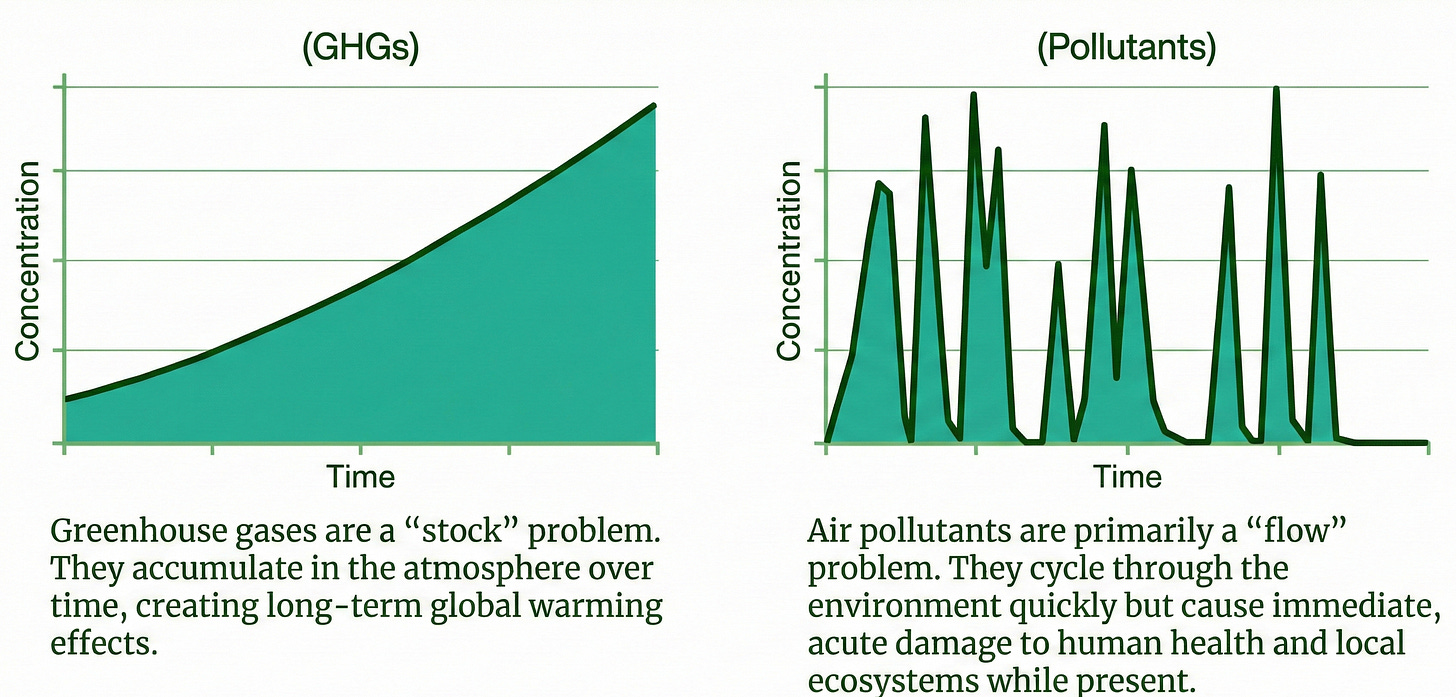
In the corporate sustainability conversation, we often lump emissions into one category. However, it is vital to distinguish between Greenhouse Gases (GHGs) and air pollutants. While often described as “two sides of the same coin,” they behave differently:
Greenhouse Gases (CO2, Methane, etc.): These are a global stock problem. They accumulate in the atmosphere over centuries and primarily affect the climate by trapping heat.
Air pollutants (PM2.5, NOx, Ozone, etc.): These are a local and regional flow problem. They remain in the air for a short time (days to weeks) but cause immediate damage to human health and ecosystems.
The major air pollutants
To understand the risk, we must look beyond just carbon. The primary pollutants include:
Particulate Matter (PM2.5 and PM10): Tiny solid or liquid particles suspended in the air. PM2.5 is particularly dangerous because it is small enough to penetrate deep into the lungs and enter the bloodstream, causing systemic inflammation.
Black Carbon (Soot): A component of PM formed by incomplete combustion. It is a short-lived climate pollutant that hits twice: it warms the atmosphere by absorbing sunlight and, when deposited on snow or ice, it darkens the surface and accelerates melting.
Ground-level Ozone (O3): Unlike other pollutants, this is not emitted directly. It is a secondary pollutant formed when sunlight reacts with chemical ingredients (NOx and VOCs) in the air. It is the main component of smog and is highly toxic to human lungs and crops.
Nitrogen Oxides (NOx) and Sulfur Dioxide (SO2): Reactive gases formed primarily from burning fossil fuels. They are major lung irritants and the primary drivers of acid rain.
Volatile Organic Compounds (VOCs): Chemicals that vaporize easily (found in paints, solvents, and fuels). While some are directly toxic, their main danger is acting as a precursor for ground-level ozone.
Ammonia (NH3): Often overlooked in industrial reports, this gas primarily comes from agriculture (fertilizers and livestock). It reacts with NOx and SO2 in the air to form dangerous secondary particulate matter.
The transboundary issue
We tend to think of pollution as a localized urban issue—smog over a city. However, phenomena like Atmospheric Brown Clouds (ABCs) prove otherwise. These are vast, 3-kilometer-thick layers of pollution consisting of aerosols, soot, and chemical particles that can span entire ocean basins. They prove that air pollution is not just a city problem; it is a transcontinental crisis that alters weather patterns and agriculture thousands of kilometers from the source.
3. Business as the driver of air pollution
Incomplete combustion (energy & heat)
The most significant source of industrial air pollution is the burning of fossil fuels (coal, oil, natural gas) and biomass.
The mechanism: When fuel is burned to generate electricity, power boilers, or run heavy machinery, it rarely burns “cleanly.”
The output: This releases Carbon Dioxide (CO2), but also Nitrogen Oxides (NOx), Sulfur Dioxide (SO2), and Black Carbon.
Key sectors: Energy generation, heavy manufacturing, and heating.
Industrial processes & fugitive emissions
Many industries release pollutants from the chemical reactions inherent to their production processes. And not just from burning fuel for power.
Iron and steel: This industry is a major source of dust and heavy metals. The sintering and coking processes release vast amounts of PM, SO2, and NOx.
Chemicals and solvents: Industries using solvents (paints, coatings, cleaning agents) release VOCs through evaporation. These are known as fugitive emissions because they often leak from valves, pumps, or open vats rather than a smokestack.
Construction & mining: These sectors are responsible for significant coarse particulate matter (dust) generated by breaking rocks, moving soil, and demolition.
Supply chain & logistics (the hidden pollution)
For many companies, the bulk of their air pollution footprint lies outside their own fence line, within their supply chain.
Transportation: The global movement of goods relies heavily on diesel engines (trucks, ships, trains). Diesel exhaust is a primary source of NOx and Particulate Matter.
The “Pollution Haven” effect: There is a phenomenon known as the “Pollution Haven Hypothesis,” where multinational corporations may relocate dirty production stages to developing countries with laxer environmental regulations. This effectively offshores the air pollution, allowing the company to appear cleaner at home while contributing to global atmospheric degradation (such as the Asian Brown Cloud).
The Asian Brown Cloud is a massive, three-kilometer-thick layer of air pollution that hangs over South Asia and the Indian Ocean, primarily during the dry winter months. It is caused by a mixture of soot, dust, and chemicals from burning fossil fuels in factories and vehicles, as well as the burning of wood and crop waste in rural areas. Because there is no rain to wash the air clean between November and April, this toxic haze builds up and traps heat while blocking sunlight, which disrupts weather patterns, accelerates the melting of Himalayan glaciers, and poses serious respiratory health risks to millions of people.
Sources
The Atmosphere: Getting a Handle on Carbon Dioxide - NASA Science
Frontiers | Environmental and Health Impacts of Air Pollution: A Review
Editorial: Atmospheric chemistry and air pollution - PMC
Global supply chain relocation reshaped PM2.5 pollution and human health risks